The cost-effectiveness of thrombopoietin mimetics varies substantially in patients with thrombocytopenia
07/06/23
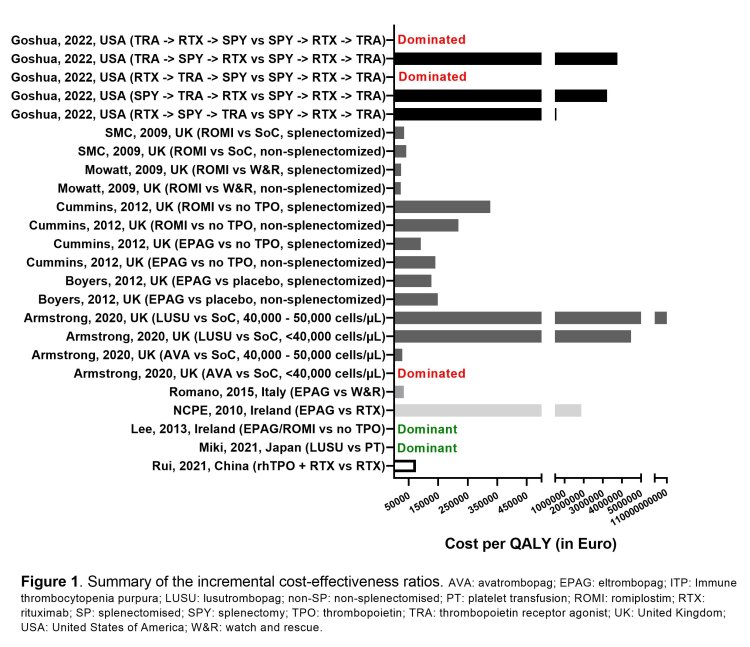
Thrombopoietin (TPO) mimetics are potential alternatives to platelet transfusion for reducing blood loss in patients with thrombocytopenia (low platelet count). Based on an identified evidence gap in a previously developed scoping review on existing systematic reviews in the field of platelet transfusion, we conducted a systematic review to identify full economic evaluations and randomized controlled trials assessing the cost-effectiveness of TPO mimetics (e.g. romiplostim, avatrombopag, eltrombopag, lusutrombopag) compared to platelet transfusions or other first-line (corticosteroids or intravenous immunoglobulins) or second-line treatment modalities (immunosuppressants, splenectomy, rituximab, danazol).
Eight databases and registries were searched, resulting in 20 full economic evaluations and 1 randomized controlled trial, which showed that the cost-effectiveness of TPO mimetics, in terms of the incremental cost per quality-adjusted life year (QALY), ranged from a dominant strategy (i.e. cost-saving and clinically superior), to a significant incremental cost per QALY, or a dominated strategy (i.e. increased costs and clinically inferior), see Figure 1.
Future validation and tackling the uncertainty of these models with country-specific cost data and up-to-date efficacy, safety, and utility data are needed to increase the generalizability of the current cost-effectiveness estimates and to use these data in international evidence-based guideline initiatives.
This systematic review has been published in Pharmacoeconomics and can be found here.
